You often hear me talk about pH but from your messages I understood that there is often confusion about it and it is not always clear what it is for and how we can use it in henna and herbal dye mixes to obtain a color that is close to our needs .
So it is necessary to talk about a bit of chemistry to know the pH and how it can affect the tone of the reflection released by the vegetable pigment of henna and dyeing herbs on our hair.
What is pH?
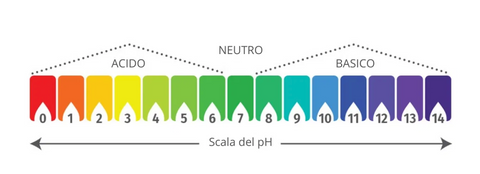
Let's start from the premise that pH is a scale for measuring the acidity or basicity of an aqueous solution , so remember that it can only be measured if the substance is soluble in water.
It has a reference scale that goes from 0 (maximum acidity) to 14 (maximum basicity) and where the value 7 indicates neutrality.
So all solutions with a pH below 7 are ACID while those above 7 are BASIC.
But if you can measure the pH of water-soluble substances why do we talk about the pH of the skin or the pH of the hair if they are not soluble in water?
On the hair and on the skin there is always a light hydrolipidic, protective-moisturizing mantle composed of sweat (thanks to the sweat glands) and sebum (thanks to the sebaceous glands).
The sebum produced by the sebaceous glands has a precise role, not only in contributing to the formation of the hydrolipidic film on the skin, but also has the function of lubricating the hair as it grows upwards.
So the hydrolipidic mantle made up of 70% water and 30% fatty and mineral substances is an organic solution, naturally produced by our body with a physiological pH value of 4.5-5.5.
The natural pH of dyeing herbs
Some herbal dyes prefer a basic pH rather than an acidic pH to obtain the maximum dyeing performance on the hair.
For example:
Indigo and katam naturally develop a basic pH when water is added to the dye powder.
So to obtain the maximum dyeing yield from the vegetable pigment released by Katam or Indigo we must favor the creation of a basic environment and not contrast it with ingredients that can make the environment acidic.
For example , using Amla and Katam or Indigo in the same mix could prove counterproductive if you aim for a dark and intense color because the pH of Amla is very acidic and therefore prevents the dye mix from maintaining an alkaline pH.
There are those who prefer to add a pinch of baking soda to emphasize the dyeing power and reflections of these 2 dyeing powders but I encourage you to always experiment firsthand and decide whether or not you need to add a pinch of baking soda into your mix.
Instead the pH of lawsonia is naturally acidic . Also in this case there are those who add lemon juice or a few drops of vinegar - but also in this case they always experiment firsthand.
Other reflexing herbs totally change their reflex according to the pH of the environment in which they are found, such as turmeric, rhubarb, madder or hibiscus.
In this article, you can see how the hue obtained with turmeric or rhubarb varies based on the basic or acidic environment in which it was released.
How can I obtain a BASIC or ACID pH?
To create a dyeing batter from an acid or basic environment at home, you can use substances that you find in your kitchen:
- lemon or vinegar to create ACID solutions / environments
- bicarbonate to create BASIC solutions / environment
To create a basic or acidic pH for our dyeing mixture, just use a very minimal quantity, a knife tip of bicarbonate or the juice of half a lemon.
The alkaline pH acts on the stem by swelling it by opening its scales to allow the chemical substances to penetrate inside and modify its structure.
If the alkaline environment has a pH that approaches the maximum basicity ( 13-14 ) the dilatation on the stem is so strong as to irreparably damage the hair.
The acid pH instead closes the open scales of the stem. Usually these are treatments (conditioner, acid rinse or styling spray such as the Volumizing or Anti-frizz spray with fruit acids) that are used on the hair following basic pH treatments (chemical coloring or simply a slightly alkaline pH shampoo such as with chickpea flour or a dyeing mix based on katam and indigo).
Shampoos usually have a pH that varies between a pH of 4.5 - 5, while treating shampoos such as Shampoo Scrub reach a pH of 6 which, compared to the physiological pH of the scalp and hair, can be basic.
A good conditioner has the role of conditioning the hair but also closing the scales thanks to its acidic pH . They usually have a pH that can vary between a value of 3-4.
Obviously, you must always use acidic substances consciously and not abuse them because hair subjected to substances whose value is close to pH 1, suffers irreparable damage.
To conclude, I leave you with a list of various substances and their pH which I am sure you will find curious in some cases:
- Citric acid, lemon juice pH 2.0/2.4
- Vinegar pH 2.9
- Oxygen for hair coloring pH 3.0/3.5
- Conditioners and restructuring masks pH 4.0/5.0
- Sweat pH 4.8/6.6
- Swimming pool water pH 7.2/7.8
- Seawater pH 7.7/8.4
- Alkaline soaps pH 9.0/10.0
- Hair dye pH 9.00/11
- Hair bleaches pH 10.5/11.5
- Ammonia pH 12.0/13.0
- Sodium hydroxide ( caustic soda ) pH 14
© Beautiful Delights
In the online shop you will find everything you need (including herbal dyes) to take care of your skin and hair in a conscious way. Click HERE to visit it!
************************************************** ************************************
Keep in touch with us:
Follow us on SOCIAL by clicking on the icons below to stay up to date on the latest news:
Rozalia & the Beautilicious Delights Team.




